- Company
About our company
Our goal since 2014 has been to bring more human milk to babies. Learn more about how our purpose-driven approach allows us to rethink human milk processing from the ground up. - A new standard of care
A new standard of care
We’ve reimagined what infant nutrition can look like. From the NICU to at-home care, every baby will be able to have access to the life-saving benefits of human milk.
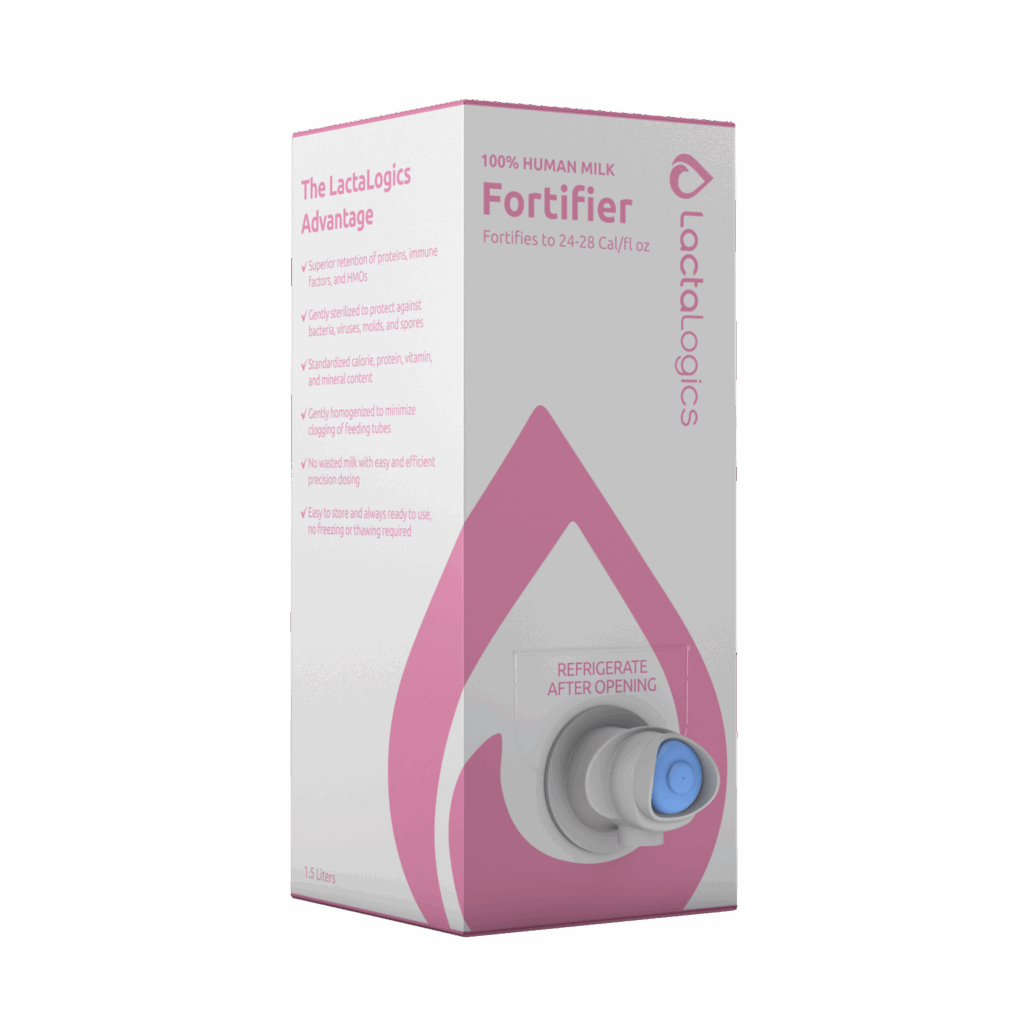
- Human milk products
Human milk products
We aim to provide babies with a reliable, safe, and abundant source of human milk. Learn more about our 100% human milk-based fortifier and ready-to-feed products. - Hub
Clinical Hub
A curated collection of peer-reviewed research, clinical resources, press coverage, and educational materials on human milk-based nutrition for premature and at-risk infants.
- For donors
For donors
We’re connecting moms who have healthy babies and can produce excess breast milk to babies in need. Help give babies the nutrition only human milk can give.